General Principles of Thermal Lensing
Thermal lens spectrometry appeared in practical analysis in the beginning of the 1980`s and proved to be a powerful tool of trace analysis. Like other thermo-optical methods, thermal lens spectrometry is based on the effect of a change in thermo-optical properties of a sample illuminated by a laser light source owing to absorption of the energy of radiation. Measurements of the change in divergence of a laser beam after formation of the thermal lens allows determination of the absorbances of 10-7 – 10-6, which correspond to analyte concentrations of 10-11 – 10-10 mol/L. Thus, thermal lens spectrometry is 100 — 1000 times more sensitive than conventional spectrophotometry.
Back to the top...The scheme of analytical signal formation in thermal lens spectrometry (for single beam measurements):
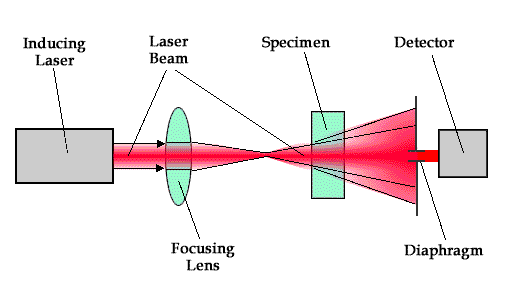
The size of the laser beam before and after the formation of a thermal lens are marked by the line.
Back to the top...
Thermal lens spectroscopy appeared as an analytical method twenty five years ago. Twenty five years for an analytical method is a rather considerable period. Such a test of time is enough to figure out the place of thermal lens spectrometry among other methods and to reveal the prospects of its development. Thermal lens spectrometry has with some problems caused by sophisticated laser equipment, lack of serial devices, high requirements for the personnel and a competition of other highly sensitive methods. However, despite these problems thermal lens spectrometry consolidates its positions, which is viable itself.
An important feature of the current status of thermal lens spectrometry is a change of the majority of examinations from the "reproduction" of spectrometric procedures to a search for the new approaches that are based on thermooptical procedures. The most known developments deal with the development of the photochemical thermal-lens spectrometry, detection in capillary electrophoresis, high-performance liquid chromatography, etc.
After twenty five years of the development, thermooptical spectrometry has passed a long way, and the interest to the methods in question was not always was constant (see the figure).
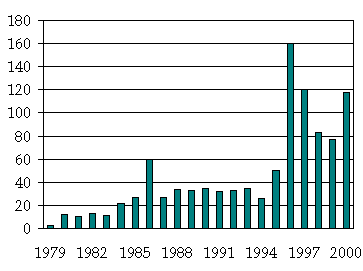
The number of papers on thermooptical spectrometry in analytical and related journals.
The development of the method experienced three stages; and the quality level of studies and their analytical value grew from stage to stage. Let us try to characterize briefly each direction of development of the method. First, the most widespread and simple variant of analytical applications of thermooptical spectrometry consists in a simple transfer of existing spectrophotometric procedures to the conditions of thermooptical detection. Measurements of a change in the divergence of a laser beam after the blooming of the thermal lens allows determination of the absorbances of 10–7 – 10–6, which correspond to analyzed concentrations of 10–11 – 10–10 molЧL–1. Thus, thermal lens spectrometry is 100 — 1000 times more sensitive than conventional spectrophotometry. A great advantage of TLS is that it can easily apply any procedures based on conventional spectrophotometric techniques. Thermal-lens detection allows an increase in the sensitivity of existing spectrophotometric procedures by 1-2 orders.
During a long period of time this direction was considered most promising, as it allowed the researchers to combine reliable procedures based on conventional spectrophotometric techniques and high instrumental sensitivity of thermooptical spectrometers. Nowadays, three quarters of thermooptical procedures are based on this approach. In fact, it is rather fruitful for measuring impurities in natural samples and in the case when a decrease in the limit of detection is restricted to the instrumental performance of the spectrometer.
However, soon it has been found that a large number of facts show that it is impossible to apply existing procedures "blindly" to concentrations 2-4 orders lower than those for which they were developed. Apart from a serious problem of reagent purity, it is necessary to take into account the requirements of new and complicated secondary processes. These facts have prompted the necessity of updating(adapting) spectrometric procedures. And this was the second stage of the development of thermal lens spectrometry. It was in the full swing in the mid-1980s'. The basic aim of studies in this direction was to find the conditions providing a maximal analytical signal by updating both the chemical system and the instrumental characteristic of the spectrometer.
The third direction of the development of analytical applications of thermooptical spectrometry is the newest and, in our opinion, the most promising approach. It consists in the development of unique procedures (not necessarily photometric) using the wide variety of effects of laser radiation and also thermooptical effects on the test system. The progress in this direction is connected with chromatographic, kinetic, electrophoretic and even non-analytical applications (for example, precise measurement of flow rates or the size of colloid particles).
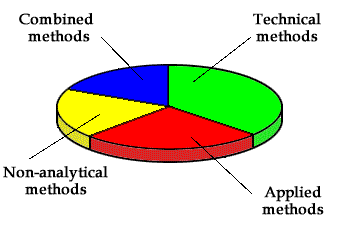
Earlier, some articles in scientific journals on analytical chemistry dealing with the technical aspects of the methods (i.e. devoted to instrumental features, mathematical processing, etc.) were dominant (see the figure).
THE DISTRIBUTION OF RESEACH PAPERS IN THERMAL LENS SPECTROSCOPY IN 1986
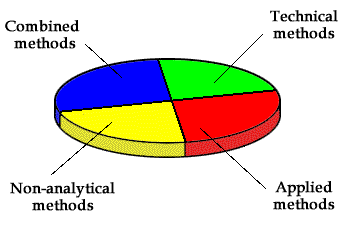
In 1997, three quarters of applications dealt with the above-mentioned specific features of thermooptical spectrometry.
THE DISTRIBUTION OF RESEACH PAPERS IN THERMAL LENS SPECTROSCOPY IN 1997
Back to the top...
- When dealing with thermal lens spectrometry, it is
possible to select six basic groups of its performance characteristics:
- High sensitivity
- Molecular absorption measurements
- Calorimetric measurements
- Power-based method and a geometrical method
- Application of lasers
- Specific features
The second group involved the performance characteristics of thermal lens spectrometry as a method of molecular absorption spectroscopy. The advantage is a large number of test substances (including organic), the possibility of operation with substances in any state, well-developed methodology, and variety of sample-preparation techniques. The important feature of a thermooptical spectroscopy is non-destructivity of determination that provides the analysis of biological objects, the remote analysis and on-line determination in the flow. The deficiencies are inherited from classical spectrophotometry, first of all, spectral nonselectivity of the method.
The parameters of thermal lens spectrometry as a calorimetric method are related to the third group of the performance characteristics. The main feature of these methods is the information about the composition and properties of the analyzed sample on the basis of measuring the absorbed radiation and the corresponding changes in physical or thermodynamic parameters. The greatest number of detectors for measuring absorbed energy (ultrasonic, interferometric, refractometric, optical, thermometrical, direct calorimetric or ex-strain-measuring (change of the sizes of a sample) are the cause of advanced calorimetric methods.
The fourth group consists of the performance parameters of a thermal lens spectrometry as a power-based method (the signal is proportional to light intensity) and a geometrical method (the signal depends on geometry of the optical scheme design).
The fifth of the basic features of a thermal lens spectrometry is interlinked with the application of lasers as intense sources of radiation. As a result, the number of effects caused by interaction of radiation with an absorbing medium sharply grows. Apart from a strong heating of the sample we deal with photochemical effects (which can play an important role as both positive and negative factors), simultaneous display of thermooptical effect, luminescence, and other effects.
At last, the sixth group includes unique properties of thermal lens spectrometry as an analytical method. Here, it is possible to point out some important moments:
Particular dimensional distribution of energy in an incident beam (mode composition). Owing to a regularity of this allocation of a profile of refractive index for the apparent thermal optics effect, it is possible to construct precise mathematical models of the effect (for example focal distance of thermal lens).
Under heating, such parameters of medium, as refractive index, density, thermal conductivity and heat capacity change. Therefore, the role of the solvent in thermal lensing is crucial. Studies of media for thermal lensing always attracted major attention. It was found, that the aqueous solutions are not optimal because of small change in the refractive index with temperature and high thermal conductivity. From this point of view non-polar solvents (benzene, tetrachlorocarbon, saturated hydrocarbons, etc.) appear most suitable for thermal lens measurements. However, the most encountered solvent in TLS is still water. Although thermooptical properties of water can be improved by increasing the temperature and adding electrolytes.
Thermooptical effects took place not in the whole sample volume, but only in a small part in the vicinity of the beam of the inducing laser (i.e. locally). A consequence is the relatively strong convection of the liquid, which can result in a blurring or periodic oscillations of the signal. However, on the other hand, the period of convective oscillations can give an important information on thermooptical properties of the test solutions. Another consequence of the locality of thermooptical effects is that chemical processes in the heated part of the solution can be an accelerated or inhibited. This fact results in the change of a rate of concentration and diffusion flows of substances in the test solution.


